Bane and boon of oxygen mediating the performance of nickel catalysts in dry reforming of methane
Catalysis is one of the key technologies in the chemical industry and has a wide-reaching impact on various aspects of our daily lives, including plastics manufacturing, drug synthesis, and production of both fertilizers and fuels. It is estimated that over 90% of chemical products are nowadays manufactured with the involvement of catalysis in at least one stage (Catal. Today, 2011, 163(1)). Catalysis is a complex process that relies on the precise structural control of several elements at the crossroads of phase (in-)stabilities. While long-term stable catalysts are indispensable in order to promote high-performance and efficient reactions, reactants undergo major chemical changes, leading to the formation of final and desired products. In heterogeneous catalysis, catalyst and reactants exist in different phases.
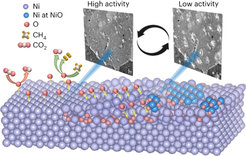
Among the various heterogeneous catalytic processes, dry reforming of methane (DRM) has recently become the subject of academic attention, as it converts two greenhouse gases, methane (CH4) and carbon dioxide (CO2), into hydrogen (H2) and carbon monoxide (CO). This mixture is also known as syngas and can be used to reduce the reliance on fossil fuels by consecutively building up larger hydrocarbons via the Fischer-Tropsch chemistry. Although Nickel- and Cobalt-based catalysts, being low cost and highly available on Earth, have shown promising activity for DRM, designing high-performance catalysts is often challenging as the connection between chemical dynamics, the formation of the active surface species and their reaction pathways is generally missing. This knowledge can only be gained from so called operando experiments in which structure and function are probed simultaneously.
A collaborative effort by scientists from the Departments of Inorganic Chemistry and Theory at the Fritz Haber Institute of the Max Planck Society in Berlin provided fundamental insights into the processes occurring at the catalyst surface and on how this modulates the catalytic performance during DRM.
In particular, they studied the role of different oxygen species on a Nickel catalyst during DRM by using a combination of experimental and computational science techniques, including operando scanning electron microscopy, near ambient pressure X-ray photoelectron spectroscopy, and computer vision. They highlighted the critical role of dissociative CO2 adsorption in regulating the oxygen content of the catalyst and CH4 activation. Furthermore, they discovered the presence of three metastable oxygen species at the catalyst: atomic surface oxygen, subsurface oxygen, and bulk NiOx. Interestingly, these exhibited different catalytic properties and their interplay and transformation gave rise to oscillations in the surface states and in the catalytic function.
They observed that some of the surface oxygen leaked into the catalyst bulk, reducing the availability of the catalyst for CH4 activation and favoring CO2 and O diffusion instead. The extent of the leakage was further proved by X-ray spectroscopy and transmission electron microscopy, revealing the presence of oxygen several nanometers below the surface of the catalysts. Consequently, new metallic sites were exposed, thus leading to an increased rate of the oxygen uptake and to a decrease of the H2/CO product ratio. Lastly, they understood that co-feeding of CO2 is essential for CH4 conversion, likely assisting its activation together with the presence of oxygenated species. "It was impressive to see how the metastability of the Ni-O system self-adjusts the catalytic performance and that one element from the reactants can direct the entire process which depends on its location and its chemistry. We hope that our findings can give new momentum in adjusting longevity and selectivity in catalysis,” says PD Dr. Thomas Lunkenbein, leader of the project and co-author of the study.
Understanding the metastability of the surfaces of catalysts, alongside with how to control them to stabilize the dynamical active state, holds important implications for the future of catalysis. In particular, it provides insights that can be transferred to the industrial level and the design of reactors where an active state with minimal energetic compromises is favored. This could be achieved either by using more potent oxidants, such as water (H2O) and nitrous oxide (N2O), or by working on reducing the oxygen leakage into the bulk by means of nanoparticles or thin film technology. The development of catalysts based on tailor-made thin films is the focus of CatLab, a joint research platform between the FHI, Helmholtz Center of Berlin (HZB) and industrial partners, which is funded by the Federal Ministry of Education and Research (BMBF) intended as a bridge between research and industry.












