New Understanding of Water Electrolysis
Water electrolysis is a key technology to establish CO2-neutral hydrogen production. One of the key technological hurdles is the design of stable, active and affordable catalysts for the anodic oxygen evolution reaction (OER), which is one bottleneck of the hydrogen production process. Researchers from the Interface Science Department have now provided quantitative near-surface structural insights into oxygen-evolving CoOx(OH)y nanoparticles which are published in Nature Energy today.

Water electrolysis is a key technology to establish CO2-neutral hydrogen production using directly the electrical energy generated by renewable power sources from sun and wind. One of the key technological hurdles is the design of stable, active and affordable catalysts for the anodic oxygen evolution reaction (OER), which is one bottleneck of the hydrogen production process. Nonetheless, so far, the near-surface structure of the working electrocatalysts is still largely unknown and thus, key catalytic properties of the ideal catalyst on the atomic scale. This lack of understanding hampers knowledge-driven catalyst optimization. Researchers from the Interface Science Department at the Fritz Haber Institute under the lead of Dr. Arno Bergmann and Prof. Beatriz Roldan Cuenya, in close collaboration with Dr. Travis Jones (AC Department), have now provided quantitative near-surface structural insights into oxygen-evolving CoOx(OH)y nanoparticles which are published in Nature Energy. Their findings on the predominant surface-terminating motif in alkali water electrolysis emphasize that the long-standing view of a large fraction in the scientific community that involved high-valent metal ions driving the OER must be reconsidered.
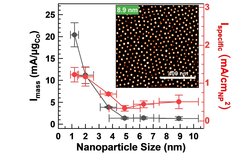
The fossil fuel-free production of green hydrogen through water electrolysis has been the topic of intense research efforts for several decades. Only recently, following late political developments as well as the decreasing cost of electricity from renewable power sources, and the possibility to couple electrolyzers directly with clean energy sources, green hydrogen has become economically appealing. In water electrolysis, the anodic oxygen evolution reaction (OER) remains the bottleneck, which so far limits the overall efficiency and hinders broader industrial application. In catalyst development, the nanoparticle size is a critical parameter which minimizes the use of material. The investigation of size-dependent properties and their catalytic function can provide crucial insights into the most important properties of active catalysts.
Following this strategy, the researchers tracked the size-dependent catalytic activity of CoOx(OH)y nanoparticles down to 1 nm size, as well as their structural adaptation to OER conditions. They revealed that the smaller CoOx(OH)y nanoparticles, in particular below 5 nm, more efficiently catalyze the water electrolysis. Notably, this increase in catalytic activity was observed not only with respect to the deployed catalyst mass, ensuring more efficient material usage, but also referring to the catalyst surface area, which suggest an intrinsic size effect related to the increased abundance of highly active surface sites. These findings provide strategies to design improved catalysts for alkaline water electrolysis, which are now ready to be tested under industrial conditions.
Furthermore, the researchers also revealed a size-dependent change in the structure and chemical state of the CoOx(OH)y nanoparticles during the alkaline oxygen evolution reaction, leading to a near-surface Co-O bond contraction. Here, the outstanding methodical experimental capabilities and interdepartmental collaboration at the Fritz Institute enabled to show that the CoOx(OH)y reversibly accumulate charge in the near-surface under OER conditions, which is only detectably by operando X-ray absorption analysis. This charge accumulation is not only driving catalytic processes, but also inducing an oxidation and contraction of the Co-O bonds. Comparing the experimental insights with theoretical calculations from the AC Department allowed us to locate the accumulated oxidative charge at the oxygens on the catalyst surface, leading to the presence of an oxyl-adsorbate. This contrasts Co4+ formation previously suggested in the literature and unveils charge reorganization in the 3d orbitals of the Co3+ ions during OER. Most importantly, the researchers showed that the average charge accumulation increased for the smaller and more active nanoparticles, yet to a lesser degree than expected considering the surface to-volume ratio. Thus, they were able to extract first quantitative insights into the near-surface structural adaptation of catalysts to the OER conditions by analyzing the size-dependent changes. Finally, their combined findings on the catalytic function and the properties of the working catalyst greatly contribute to draw an unified operando mechanistic understanding of OER, a prerequisite of improved knowledge-driven electrocatalyst design for alkaline electrolysis and thus, more efficient hydrogen production.













